-
PDF
- Split View
-
Views
-
Cite
Cite
Felix Sommer, Satoko Awazu, Friederike Anton-Erxleben, Di Jiang, Alexander V. Klimovich, Boris V. Klimovich, Marina P. Samoi˘lovich, Yutaka Satou, Margret Krüss, Christoph Gelhaus, Ulrich Kürn, Thomas C.G. Bosch, Konstantin Khalturin, Blood System Formation in the Urochordate Ciona intestinalis Requires the Variable Receptor vCRL1, Molecular Biology and Evolution, Volume 29, Issue 10, October 2012, Pages 3081–3093, https://doi.org/10.1093/molbev/mss120
Close - Share Icon Share
Abstract
Adaptive immune systems are present only in vertebrates. How do all the remaining animals withstand continuous attacks of permanently evolving pathogens? Even in the absence of adaptive immunity, every organism must be able to unambiguously distinguish “self” cells from any imaginable “nonself.” Here, we analyzed the function of highly polymorphic gene vCRL1, which is expressed in follicle and blood cells of Ciona intestinalis, pointing to possible recognition roles either during fertilization or in immune reactions. By using segregation analysis, we demonstrate that vCRL1 locus is not involved in the control of self-sterility. Interestingly, genetic knockdown of vCRL1 in all tissues or specifically in hemocytes results in a drastic developmental arrest during metamorphosis exactly when blood system formation in Ciona normally occurs. Our data demonstrate that vCRL1 gene might be essential for the establishment of a functional blood system in Ciona. Presumably, presence of the vCRL1 receptor on the surface of blood cells renders them as self, whereas any cell lacking it is referred to as nonself and will be consequently destroyed. We propose that individual-specific receptor vCRL1 might be utilized to facilitate somatic self/nonself discrimination.
Introduction
Human-like adaptive immune system and histocompatibility are not present outside jawed vertebrates. The only other example of somatically diversified blood cell receptors that also allow to recognize unlimited variety of antigens are variable lymphocyte receptors of lamprey and hagfish (Pancer et al. 2004, 2005; Boehm et al. 2011). Thus, adaptive immune system seems to be restricted to vertebrates. How do the animals lacking it succeed to withstand continuous attacks of permanently evolving pathogens? It is clear that even without adaptive immunity, each organism must be able to unambiguously distinguish “self” cells from any imaginable “nonself.” In invertebrates, however, functional and evolutionary connections between immunity and self/nonself discrimination still remain obscure.
Urochordates, being the closest living relatives of vertebrates (fig. 1A; Delsuc et al. 2006, 2008), represent an ideal model system to gain insights into the evolution of self/nonself recognition systems. Compound ascidians such as Botryllus are well known for naturally occurring colony fusion/rejection reactions (Bancroft 1903; Oka and Watanabe 1957). Solitary ascidians are hermaphrodites and possess a block of self-fertilization (Morgan 1923; Fuke 1983). Thus, urochordates simultaneously utilize two types of self/nonself discrimination systems: tissue histocompatibility and self-sterility. Accordingly, several highly polymorphic and individual-specific receptors have been isolated from urochordates (fig. 1A). In Botryllus schlosseri, two genes responsible for colony fusion/rejection have been identified within the Fu/HC (fusibility/histocompatibility) locus (De Tomaso et al. 2005; Nyholm et al. 2006). In Halocynthia roretzi and H. aurantium, HrVC70 and HaVC80 proteins were shown to be potential self-sterility determinants (Sawada et al. 2004; Ban et al. 2005). In Ciona intestinalis, three highly polymorphic loci, Themis-A, Themis-B, and vCRL1, have been reported (Kürn et al. 2007; Harada and Sawada 2008; Harada et al. 2008). Themis loci are responsible for the block of self-sterility in Ciona subspecies A (Western and Eastern Pacific). vCRL1 had been identified in unbiased screening for variable genes expressed in gonads of Ciona subspecies B (from North Atlantic) and proposed to be involved in the prevention of self-fertilization (Kürn et al. 2007).
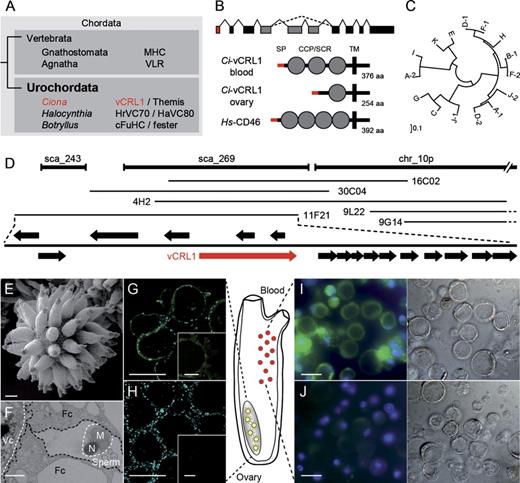
The vCRL1 protein is located on the surface of oocytes and hemocytes. (A) Chordate phylogeny and genes involved in self/nonself recognition processes. (B) Domain organization of vCRL1 protein. (C) vCRL1 is individual specific and highly polymorphic as revealed by neighbor-joining phylogenetic analysis of ovarian proteins. vCRL1 protein sequences were deduced from full-length transcripts. Letters indicate proteins from the individual animal and numbers designate allelic variants. Protein alignment is shown in supplementary figure S1 (Supplementary Material online). (D) Schematic representation of isolated BACs containing the vCRL1 locus. Scaffolds and clones are drawn in scale. Arrows represent predicted open reading frames. (E) SEM micrograph of spawned mature oocyte. (F) Transmission electron microscopy picture of mature oocytes incubated with heterologous sperm. Note the diameter of the sperm head is 1–2 μm and therefore larger than the ridge between two follicle cells. The follicle cell surface as well as the sperm head has been highlighted using dashed lines for better visualization. Fc—follicle cell, Vc—vitelline envelope, N—nucleus, M—mitochondrion. (G–J) Localization of vCRL1 protein by immunohistological staining using ovary sections (G, H), spawned mature oocytes (inlets in G, H) and hemocytes (I, J). Tissues were incubated with α-vCRL1 (G, I) or control serum (H, J). For hemocytes, bright-field photographs are shown on the right. Blue–Hoechst stained nuclei, green—anti-mouse Alexa 488 secondary antibody. Bars represent 20 (E), 1 (F), 50 (G, H), and 5 μm (I, J).
In this study, we analyzed the function of vCRL1 gene in C. intestinalis. Its intriguing feature is that each Ciona individual in a wild population possesses two unique allelic variants, which are not shared with any other individual unless they are closely genetically related. vCRL1 encodes a type I transmembrane protein with several short consensus repeat/complement control protein (SCR/CCP) domains (fig. 1B), thereby structurally resembling complement receptors of the human regulators of complement activation gene cluster (e.g., CD46, CD55). vCRL1 is expressed in follicle cells covering mature oocytes and also in blood cells, pointing to possible roles in fertilization or in immune reactions (Kürn et al. 2007).
Here, we used anti-vCRL1 antibodies, segregation analysis, and transgenic knockdown animals to examine the involvement of vCRL1 locus in self/nonself discrimination processes in subspecies B of C. intestinalis (supplementary fig. S1, Supplementary Material online). Our data show that vCRL1 is not involved in prevention of self-fertilization. Interestingly, knockdown of vCRL1 using hairpin constructs driven by ubiquitous (EF1α) promoter blocks metamorphosis at the time point when blood system formation in Ciona normally takes place. Moreover, knockdown of vCRL1 expression specifically in blood cells by hemocyte-specific vWA-like promoter also represses metamorphosis, decreases the number of hemocytes, and prevents differentiation of circulating blood cells into tunic cells. Thus, vCRL1 seems to be essential for the establishment of a functional blood system in Ciona.
In the Ciona genome, vCRL1 gene is located at the short arm of chromosome 10. This region is particularly interesting as it simultaneously harbors multiple genes with CCP core elements typical for proteins regulating complement system and receptors belonging to immunoglobulin superfamily (IgSF). It has been already proposed that 10q region of the Ciona genome has similarities to the leukocyte receptor complex of vertebrates and might be important for the immune functions of the hemocytes (Zucchetti et al. 2009). Based on our results, we hypothesize that presence of vCRL1 receptor on the surface of blood cells renders them as self, whereas any cell lacking it is referred to as nonself and will be consequently destroyed. Individual-specific receptors such as vCRL1 might be utilized to facilitate self/nonself discrimination in Ciona. We propose that proteins controlling complement system might have an evolutionary ancient role not only in immune defense but also in histocompatibility.
Materials and Methods
Biological Material
Living C. intestinalis were obtained from Biological Station Helgoland (Alfred-Wegener-Institute, Germany). Animals were kept in aquaria containing North Sea water at 15 °C with constant aeration. Water was cleaned in a closed system including biofilter and protein separator. Animals were fed twice a week with Liquifry Marine (Interpet Ltd). For the segregational analysis, Ciona individuals were sampled at Bergen seaside and cultured at SARS International Centre for Marine Molecular Biology (Bergen, Norway). Animals from both locations belong to C. intestinalis subspecies B (supplementary fig. S1, Supplementary Material online).
Immunohistochemistry
Ciona intestinalis tissue was fixed with 4% (w/v) paraformaldehyde in seawater at 4 °C for 16 h and transferred to methanol. After washing with PBS with 0.1% Tween-20 (PBT), permeabilization was performed in phosphate-buffered saline (PBS) with 0.5% Triton X-100. Blocking was carried out in 1% (w/v) bovine serum albumin (BSA) in PBT and tissue incubated with primary antibody diluted 1:100 to 1:500 in blocking solution. After washing with 1% (w/v) BSA in PBT, donkey-anti-mouse Alexa 488 secondary antibody (Invitrogen; 1:1000 in blocking solution) was added. Unbound antibody was washed away with PBT and DNA staining of cell nuclei was performed using 1 μg/ml HOECHST dye. Tissue was embedded in 1,4-diazabicyclo-[2,2,2]-octane/Mowiol. Fixed tissue was stored in methanol at −80 °C.
Fertilization Experiments
Mature Ciona animals were cultured under continuous illumination for at least 48 h to inhibit spawning. Gametes were extracted directly from gonoducts after dissection. Sperm was diluted in filter-sterilized seawater (FSW) to OD600 of 1.0 just before insemination. Approximately 500 oocytes in 250 μl FSW were mixed with 10 μl auto- or heterologous sperm. After 75 min incubation at room temperature (RT), cells were fixed by addition of one volume 2 M sulphuric acid and fertilization was assessed by cleavage status. All fertilization experiments were carried out in 48-well plates with flat bottom.
Antibody Interference
Oocytes were preincubated in FSW containing different concentrations of mouse anti-vCRL1 serum or as control preimmune serum for 60 min at RT. Crossing experiments were carried out as described above.
Segregation Analysis
Seven C. intestinalis individuals were collected from a fjord (60°31′58″N, 5°25′71″E) near Bergen (Norway) and crossed in different combinations (see table 1). Sperm, ovary as well as samples of somatic tissues were preserved at −80 °C for every parental animal. Animals resulting from crosses were cultured until sexual maturity in laboratory, which took about 4 months. Siblings were checked for cross-fertilization ability against other individuals of the same cross as described above, and whole animals were preserved in 70% ethanol for preparation of genomic DNA. Synthesis of dscDNA was performed using ovary mRNAs isolated from parental animals. The complete full-length vCRL1 transcript sequences were determined by splinkerette polymerase chain reaction (PCR). Primers were designed against conserved transcript sequences coding for the CCP domain and the 3′ untranslated region (UTR). Using these primers, fragments of the vCRL1 genomic loci from parental animals were amplified and sequenced by primer walk. All vCRL1 alleles of the crosses were finally analyzed by AFLP using primers CCP_for8 GCA ACM TGT CAG GAG RAT GC and vCRL_CCP2_r CAT CAT TAC CAC ATG TAG CAG AC or Geno_rev1 GAA CCA GTT TGA ATA AAA CAA TAC CG designed against parental genomic vCRL1 sequences. To analyze Themis-A and Themis-B alleles in the crosses, primers s-Themis-A_for/rev or 151120_for/rev (Harada et al. 2008) were used.
Statistics of Performed Crosses and Hypothetical Parental Genotypes.
| P . | Crosses . | Cross-sterile . | % . | Predicted Genotype . |
|---|---|---|---|---|
| A × B | 31 | 2 | 6.5 | het/het × het/het |
| 30 | 2 | 6.7 | ||
| 61 | 4 | 6.6 | ||
| B × G | 30 | 4 | 13.3 | hom/het × het/het |
| 17 | 3 | 17.6 | ||
| 32 | 0 | 0 | ||
| 38 | 4 | 10.5 | ||
| 7 | 1 | 14.3 | ||
| 124 | 12 | 9.7 | ||
| C × D | 30 | 2 | 6.7 | hom/het × het/het |
| 30 | 4 | 13.3 | ||
| 60 | 6 | 10.0 | ||
| C × F | 36 | 8 | 22.3 | hom/het × het/het |
| 30 | 2 | 6.7 | ||
| 30 | 0 | 0 | ||
| 30 | 2 | 6.7 | ||
| 126 | 12 | 9.7 | ||
| E × F | 30 | 8 | 26.7 | hom/het × hom/het |
| 6 | 2 | 33.3 | ||
| 36 | 10 | 27.8 | ||
| Total | 407 | 44 |
| P . | Crosses . | Cross-sterile . | % . | Predicted Genotype . |
|---|---|---|---|---|
| A × B | 31 | 2 | 6.5 | het/het × het/het |
| 30 | 2 | 6.7 | ||
| 61 | 4 | 6.6 | ||
| B × G | 30 | 4 | 13.3 | hom/het × het/het |
| 17 | 3 | 17.6 | ||
| 32 | 0 | 0 | ||
| 38 | 4 | 10.5 | ||
| 7 | 1 | 14.3 | ||
| 124 | 12 | 9.7 | ||
| C × D | 30 | 2 | 6.7 | hom/het × het/het |
| 30 | 4 | 13.3 | ||
| 60 | 6 | 10.0 | ||
| C × F | 36 | 8 | 22.3 | hom/het × het/het |
| 30 | 2 | 6.7 | ||
| 30 | 0 | 0 | ||
| 30 | 2 | 6.7 | ||
| 126 | 12 | 9.7 | ||
| E × F | 30 | 8 | 26.7 | hom/het × hom/het |
| 6 | 2 | 33.3 | ||
| 36 | 10 | 27.8 | ||
| Total | 407 | 44 |
Note.—For individual crossings, see supplementary figure S6 (Supplementary Material online). P—parental animals, hom/het—homozygous and heterozygous parental loci.
Statistics of Performed Crosses and Hypothetical Parental Genotypes.
| P . | Crosses . | Cross-sterile . | % . | Predicted Genotype . |
|---|---|---|---|---|
| A × B | 31 | 2 | 6.5 | het/het × het/het |
| 30 | 2 | 6.7 | ||
| 61 | 4 | 6.6 | ||
| B × G | 30 | 4 | 13.3 | hom/het × het/het |
| 17 | 3 | 17.6 | ||
| 32 | 0 | 0 | ||
| 38 | 4 | 10.5 | ||
| 7 | 1 | 14.3 | ||
| 124 | 12 | 9.7 | ||
| C × D | 30 | 2 | 6.7 | hom/het × het/het |
| 30 | 4 | 13.3 | ||
| 60 | 6 | 10.0 | ||
| C × F | 36 | 8 | 22.3 | hom/het × het/het |
| 30 | 2 | 6.7 | ||
| 30 | 0 | 0 | ||
| 30 | 2 | 6.7 | ||
| 126 | 12 | 9.7 | ||
| E × F | 30 | 8 | 26.7 | hom/het × hom/het |
| 6 | 2 | 33.3 | ||
| 36 | 10 | 27.8 | ||
| Total | 407 | 44 |
| P . | Crosses . | Cross-sterile . | % . | Predicted Genotype . |
|---|---|---|---|---|
| A × B | 31 | 2 | 6.5 | het/het × het/het |
| 30 | 2 | 6.7 | ||
| 61 | 4 | 6.6 | ||
| B × G | 30 | 4 | 13.3 | hom/het × het/het |
| 17 | 3 | 17.6 | ||
| 32 | 0 | 0 | ||
| 38 | 4 | 10.5 | ||
| 7 | 1 | 14.3 | ||
| 124 | 12 | 9.7 | ||
| C × D | 30 | 2 | 6.7 | hom/het × het/het |
| 30 | 4 | 13.3 | ||
| 60 | 6 | 10.0 | ||
| C × F | 36 | 8 | 22.3 | hom/het × het/het |
| 30 | 2 | 6.7 | ||
| 30 | 0 | 0 | ||
| 30 | 2 | 6.7 | ||
| 126 | 12 | 9.7 | ||
| E × F | 30 | 8 | 26.7 | hom/het × hom/het |
| 6 | 2 | 33.3 | ||
| 36 | 10 | 27.8 | ||
| Total | 407 | 44 |
Note.—For individual crossings, see supplementary figure S6 (Supplementary Material online). P—parental animals, hom/het—homozygous and heterozygous parental loci.
Knockdown of vCRL1 Gene Expression
Constructs carrying vCRL1 or as control Hydra vulgaris AEP signal transmitter 1 (Hemmrich and Bosch 2008) hairpin sequences were introduced into C. intestinalis embryos by electroporation (Sasakura et al. 2007, 2008). Electroporations were performed using GenePulser XcellTM (Bio-Rad) at parameters 15 ms, 50 V, and 60 μg of construct DNA. After electroporation, embryos were cultured in sterile-filtered seawater at 14 °C in the presence of 50 μg/ml ampicillin and 50 μg/ml kanamycin. Hairpin construct was designed according to the following guidelines: 1) vCRL1 transcript sequence encoding transmembrane domain as well as 3′UTR was used as template because it shows the least polymorphism between individuals, 2) sense and antisense sequences were separated by a spacer of at least 300 nucleotides. Cloning was performed using modified pGEM-T vector (Promega) as backbone, and the assembled cassettes were transferred to pMiLRneo expression plasmid (Klinakis et al. 2000) to create the final constructs. Construct integration was monitored by EGFP reporter gene expression driven by two different promoters, the strong and ubiquitous Ciona EF1α (see expression profile in Aniseed http://aniseed-ibdm.univ-mrs.fr/) and the hemocyte-specific vWA-like promoter (Wakoh et al. 2004). Both promoters were approximately 2 kb in size and amplified using primers EF1α_P_f_Sbf AAA ACC TGC AGG GAA TGT AAC TTG CTT TAC CAT CGC G and EF1α_P_r_PacI TTT TTT AAT TAA AGT CTT GTC TTT CAT TTT GGA AGG TTG or vWA_P_for AAA ACC TGC AGG CTT TGG TTA TAG GCT ATT GTT TGA GG and vWA_P_rev TTT TTT AAT TAA TAG TAG CAG CTT CAT ATT TAA TAA CTT CAC. Approximately 1.5 kb sequence downstream of ci0100150109 (ANISEED, http://aniseed-ibdm.univ-mrs.fr/ or JGI genome browser, http://genome.jgi-psf.org/Cioin2/Cioin2.home.html) was amplified by EF1α_T_f_Asis AAA AGC GAT CGC GTG ACT GCA GCC AAC GAC AC and EF1α_T_r_Asc TTT TGG CGC GCC CAA TCT AAC TGA TAA GCA GAG TCA TG and was used as a terminator in all constructs. For knockdown experiments, only those electroporated animals were chosen, which showed more than 75% transgenic cells in the whole embryo and normal tadpole development. Tadpoles were allowed to settle in 2 ml antibiotic seawater in 24-well culture dishes with one tadpole per well. Water was changed every 2 days. Progress of metamorphosis was scored by monitoring if development of an individual failed and arrested at attachment, rotation, or first ascidian stage at the end of 12 days since planting in the well. For reverse transcription (RT)-PCR, we used the following primers: AK17utr18_F GAA TAT TCT TTG CAT GTT CCA GG and vCRL_UTR_r3 GTG ACG AAA CAA CGT TAA TTT GC for vCRL1; vWA_F CAC GTA GCG TCC CTT GCG GC and vWA_R GTC GTT GCA ACC AAT AGT TCC for vWA-like; and ciActin_F115 TAC GAG GGT TAC GAA ATT CCC CAC and ciActin_R454 TAG GTG GTC TCG TGG ATA CCA GC for beta-actin.
Molecular Techniques
Isolation of nucleic acids and proteins, cDNA synthesis, cloning, and sequencing were done following standard protocols (Sambrook et al. 1989).
Rapid Amplification of 5′ Complementary DNA Ends
Full-length cDNA sequences were amplified by splinkerette PCR as described previously (Siebert et al. 2005). Splinkerette was ligated to dscDNA 5′ends and PCR performed using primers against splinkerette sequence as well as to the nonpolymorphic 3′UTR of the vCRL1 transcript.
Production of Recombinant vCRL1 Proteins
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) was carried out according to Laemmli (1970). Samples were diluted in 2× loading buffer, boiled for 5 min, and loaded onto 10–18% polyacrylamide gels. Electrophoresis was performed at 30 mA and prestained protein marker (New England Biosciences) was used to estimate protein sizes. Proteins were visualized by staining with Coomassie. Escherichia coli Rosetta 2(DE3)pLysS cells (Novagen) were transfected with vCRL1 expression vectors according to manufacturer's protocol. Vectors were based on pET28a plasmid in which vCRL1 sequences were inserted by XhoI as well as NdeI digestion. Recombinant vCRL1 protein was extracted from 500 ml induced culture. Cells were pelleted and lysed by sonication, which was carried out ten times using 10% power and 70% cycle for 15 s on Sonopuls HD2200 with VS70T sonotrode (Bandelin). His-tagged vCRL1 proteins contained in the lysate were purified by denaturing Ni-NTA affinity chromatography (Novagen resin), renaturated during dialysis against 50 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid, pH 8.0, and concentrated by size exclusion chromatography using Vivaspin columns (Sartorius Stedim Biotech). Purified proteins were verified by matrix-assisted laser desorption ionization–time of flight.
Western Blot
Proteins separated by SDS-PAGE were transferred to PVDF membranes by semidry electroblotting with 3 mA/cm2 for 2 h. Dried membranes were reactivated in methanol, equilibrated in transfer buffer, and blocked with PBT supplemented with 3% (w/v) BSA. After overnight incubation with primary antibody, membranes were washed with PBT and incubated with secondary antibody (sheep-anti-mouse coupled to alkaline phosphatase, 1:2000; Chemicon). Antibodies were diluted in 1.5% (w/v) BSA in PBT. Unbound antibodies were washed away with PBT and membranes stained in nitro-blue tetrazolium and 5-bromo-4-chloro-3′-indolyphosphate 1:200 in 0.1M NaCl, 0.1M Tris, 50mM MgCl2, pH 9.5, and 0.1% Tween-20.
Generation of Mouse Polyclonal α-vCRL1 Serum
Mice were injected with bands of purified recombinant vCRL1 proteins excised from SDS-PAGE gels. Specificity of α-vCRL1 sera was elevated by boosting with solubilized purified proteins.
Light Microscopy
Confocal imaging was performed using Leica CLSM TCS SP/UV and Leica DC300F digital camera. Light microscopic analyses were carried out using Zeiss Axioskop 2 and Zeiss AxioCam digital camera.
Scanning Electron Microscopy
Tissue was fixed in 2.5% glutaraldehyde in FSW for 16 h at 4 °C. After washing with 75 mM cacodylate buffer for 30 min, postfixation was carried out with 1% OsO4 in 75 mM cacodylate buffer for 2 h at 4 °C. After washing with PBS supplemented with 350 mM NaCl, tissue was dehydrated in rising ethanol series (25%, 50%, 75%, and 100%) for 10 min twice each step. Oocytes were critical point dried in an ethanol–carbon dioxide mixture (BAL-TEC CPD030), sputter coated (BAL-TEC SCD050), and viewed at 10 kV using S420 scanning electron microscope (LEO, Leica).
Transmission Electron Microscopy
Samples were fixed as described for SEM. Further procedures were carried out as described previously (Siebert et al. 2008).
Accession Numbers
All sequences have been submitted to GenBank (accession numbers JF357715–JF357719 and JF419119–JF419171).
Results
Characterization of vCRL1 Locus
vCRL1 gene has been identified in an unbiased screening for individual-specific and variable genes expressed in the female gonad of C. intestinalis (Kürn et al. 2007). vCRL1 is a highly polymorphic gene—every individual in a population has its unique combination of two vCRL1 alleles (fig. 1C and supplementary fig. S2, Supplementary Material online, see also Kürn et al. 2007). According to the C. intestinalis genome assembly (JGI v2.0), vCRL1 is located in scaffold 269, which is 110 kb long (fig. 1D). However, scaffold 269 has a relatively low coverage by shotgun reads resulting in highly fragmented assembly with prolonged gaps. Chromosomal localization of vCRL1 gene as well as surrounding genes was not known. Thus, it was not clear whether vCRL1 locus is syntenic to Fu/HC locus of Botryllus or to any regions in vertebrate genomes where genes responsible for self/nonself recognition reside.
To determine vCRL1 gene structure as well as genes located in its direct proximity, we screened two C. intestinalis BAC libraries using vCRL1 as a probe. One library contained DNA from several Ciona individuals, whereas the second library was constructed from DNA of a single individual (Kobayashi et al. 2002). In total, six BAC clones containing vCRL1 gene were isolated and sequenced by shotgun approach (fig. 1D). Sequenced BAC clones represent four haplotypes derived from three different animals (fig. 1D and supplementary fig. S3Supplementary Data, Supplementary Material online). Five BACs overlap with scaffold chr_10p, indicating that vCRL1 gene is located at the short arm of the chromosome 10 (fig. 1D). Interestingly, vCRL1 gene is embedded into a dense gene cluster encoding transmembrane proteins with Ig and CCP domains (fig. 1D and supplementary fig. S3Supplementary DataSupplementary DataSupplementary Data, Supplementary Material online). According to the expressed sequence tag (EST) data, most of these genes are expressed in hemocytes and/or gonads (supplementary fig. S3Supplementary Data, Supplementary Material online). Number of genes in the cluster and their positions are variable among haplotypes (supplementary fig. S3Supplementary Data, Supplementary Material online). Southern blot analysis using genomic DNA of five unrelated Ciona individuals illustrates high polymorphism of vCRL1 genomic locus among Ciona individuals (supplementary fig. S3Supplementary Data, Supplementary Material online). As shown in supplementary figure S3Supplementary Data (Supplementary Material online), at least 13 genes encoding receptors with Ig/CCP domains are present in close vicinity to vCRL1. Taken together, comparison of several haplotypes demonstrates that vCRL1 is located in a highly dynamic genomic region, which undergoes frequent shuffling and rearrangements. Based on the order of genes and gene content, vCRL1 locus of Ciona is not syntenic to Botryllus Fu/HC locus but indeed as previously described by Zucchetti et al. (2009), this genomic region is highly enriched for genes, which encode receptors with IgSF core elements. Taking into consideration that most of these genes are supported by ESTs from hemocytes (supplementary figure S3Supplementary Data, Supplementary Material online), the idea that this region is particularly important for immune functions is highly plausible (for details, see Zucchetti et al. 2009).
vCRL1 Protein Is Localized on the Surface of Follicle Cells and Hemocytes
According to expression analysis by RT-PCR and in situ hybridization, vCRL1 transcript is present in follicle cells and in blood cells (Kürn et al. 2007). Exact localization of the vCRL1 protein, however, has not been determined. To address this issue, we produced recombinant proteins representing three different vCRL1 variants and used them to generate polyclonal anti-vCRL1 sera in mice (supplementary fig. S4, Supplementary Material online). Antisera were used to localize vCRL1 protein in sectioned Ciona gonads, spawned oocytes, and isolated blood cells (fig. 1E–J).
In C. intestinalis, oocytes are covered by an acellular layer called vitelline coat (VC) and two cellular layers, consisting of test cells and follicle cells (fig. 1E and F). VC plays an important role in sperm/egg recognition since it has been shown previously that heterologous sperm bind more firmly to the VC than autologous sperm (Rosati and de Santis 1978; De Santis et al. 1979; De Santis and Pinto 1991). The surface of a mature oocyte is completely covered by follicle cells and any direct contact of sperm with the VC is thereby mechanically prevented (fig. 1E and F). Since initial contact takes place between follicle cells and sperm, it has been proposed that vCRL1 gene, which is expressed in follicle cells, might be involved in self-sterility (Kürn et al. 2007). As shown in figure 1G and H, vCRL1 protein is localized in the cytoplasm as well as on the surface of follicle cells and on the VC both in developing and in mature oocytes. Thus, it is indeed present at the site of the first contact between sperm and egg.
Interestingly, large amount of vCRL1 protein is also present on the surface and in the cytoplasm of all types of hemocytes (fig. 1I and J). It has been proposed that hemocytes are the major immune effector cells in Ciona, which might be responsible for histocompatibility reactions (Arizza and Parrinello 2009). Taken together, the vCRL1 protein is present at all interfaces where self/nonself recognition in Ciona might occur.
Does vCRL1 Function as a Self-sterility Receptor in Ciona?
As vCRL1 protein is individual specific and is present in follicle cells covering oocytes, it might be involved in the control of self-sterility. To elucidate whether vCRL1 is mediating self-incompatibility, fertilization experiments were carried out in the presence of polyclonal α-vCRL1 or control preimmune mouse sera. Our initial expectation was that antiserum would mask vCRL1 receptor thereby promoting autologous fertilization but would not interfere with cross-fertilization.
Addition of α-vCRL1 serum drastically decreased the fertilization rate of heterologous inseminations compared with controls (supplementary fig. S5Supplementary Data, Supplementary Material online). Level of inhibition varied among the ten animals tested, most likely due to differences in antibody specificities reflecting high polymorphism of the vCRL1 variants among Ciona individuals (fig. 1C). Inhibition of fertilization is clearly dose dependent (supplementary fig. S5Supplementary Data, Supplementary Material online). Our data thereby indicate that the vCRL1 protein might be involved in the process of fertilization, but the mode of action was contrary to our expectation as the antibody did not promote self-fertilization. Observed effect could be also explained by the steric hindering of primary sperm binding to the VC prior to the allorecognition process, for example, by blocking interaction between CiVC57 and CiUrabin (Yamaguchi et al. 2011). Thus, it was not possible to draw the final conclusion concerning involvement of vCRL1 in self-sterility based solely on the antibody interference experiments. We, therefore, undertook genetic approach.
To further address whether vCRL1 is a self-sterility receptor, we compared alleles of this gene in cross-sterile and cross-fertile siblings (fig. 2). The basis for this segregation analysis is the haploid sperm hypothesis formulated by Thomas Morgan (Morgan 1944; fig. 2A; for details, see supplementary text, Supplementary Material online). In brief, it assumes that self-sterility in Ciona is controlled by two to five loci, which are highly polymorphic and independently inherited within the population. Fertilization is inhibited only if all alleles in all self-sterility loci are identical between two individuals. Thus, if vCRL1 receptor is responsible for self-sterility, then in all cross-sterile F1 siblings, both alleles of vCRL1 gene must be identical.
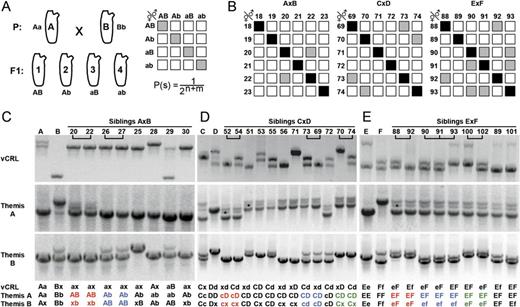
Themis-A and B but not vCRL1 alleles segregate with fertilization phenotype. (A) Basic outline of segregational analysis assuming haploid sperm hypothesis and one locus system. Progeny of parental animals A and B can have four different genotypes, therefore 16 different crosses among these F1 siblings are possible of which four are cross-sterile. The expected probability of cross-sterility can be calculated according to the depicted formula. n/m indicate the number of heterozygous parental loci. Cross-sterile and cross-fertile combinations are represented by gray and white boxes, respectively. (B) Examples of cross-sterility in performed crosses of A × B, C × D, and E × F siblings. Black—autologous controls. For full set of data, see supplementary figure S6 (Supplementary Material online) and table 1. (C–E) Segregation analysis for alleles of vCRL1, Themis-A, and Themis-B using crosses A × B (C), C × D (D), and E × F (E). Cross-sterile animals are connected with bars. Genotype of analyzed animals is given below the gel photographs in three rows representing the loci using letters according to the parents. Capital and small letters represent alleles of a heterozygote locus. If a parental allele could not be detected with the used primers, this allele was designated as “x.” Themis-A and B alleles of cross-sterile combinations are colored separately for better visualization. Unspecific amplicons for Themis-A are marked with asterisks.
Five crosses (A × B, C × D, E × F, B × G, and C × F) were produced using wild-type animals of subspecies B from Bergen, Norway (animals: A, B, C, D, E, and F). Offspring were reared to sexual maturity and crosses were set up among siblings of each batch. Cross-fertile and cross-sterile siblings were identified (supplementary fig. S6, Supplementary Material online) and their alleles in vCRL1 and previously identified self-sterility loci Themis-A and Themis-B (Harada et al. 2008) were determined (fig. 2B–E). In total, 407 crosses were performed (supplementary fig. S6, Supplementary Material online and table 1). Among all batches, we never observed frequency of cross-sterility below six percent, indicating that self-sterility in Northern European C. intestinalis, representing type B of the cryptic C. intestinalis subspecies (see supplementary fig. S6, Supplementary Material online and Suzuki et al. 2005; Caputi et al. 2007; Iannelli et al. 2007), is governed by not more than two genomic loci (table 1). The observed frequencies of cross-sterile animals of about 10% and 28% can also be explained by a two locus system. The most straightforward explanation in that case is that one or both parents are homozygous in one or both self-sterility loci, respectively (table 1).
Siblings were genotyped for vCRL1 and both Themis loci by AFLP. As shown in figure 2C–E, segregation of vCRL1 alleles in crosses C × D and E × F does not correlate with the fertilization outcomes. Several pairs of cross-sterile individuals carry different vCRL1 alleles (7 pairs of 10). This demonstrates that vCRL1 does not determine self-sterility in Ciona. Themis alleles, in contrary, perfectly correlate with the fertilization outcomes in all analyzed siblings (fig. 2C–E). Moreover, the observed hetero- and homozygotic states of Themis loci meet the expectations for parental genotypes in self-sterility loci predicted based on the crossing data frequencies (table 1). Taken together, segregation analysis demonstrates that cooperatively both Themis loci but not vCRL1 gene control self-sterility in North Atlantic C. intestinalis.
Knockdown of vCRL1 Inhibits Metamorphosis and Blood Cell Differentiation
Segregation analysis identified cross-sterile siblings, which carried different vCRL1 alleles. That unambiguously demonstrates that vCRL1 is not responsible for the regulation of self-sterility. What is then the function of this individual-specific and highly polymorphic receptor in Ciona? As shown in figure 1I, vCRL1 is expressed not only in follicle cells but also in various types of blood cells. Is this gene necessary for recognition among blood cells allowing them to distinguish each other as self and to eliminate unwanted intruders (pathogens/microbes)? To check this hypothesis, we characterized vCRL1 gene in cellular functions other than self-sterility.
As described earlier, each Ciona individual in a wild population carries its unique combination of two vCRL1 alleles. Thus, generation of transgenic animals with an additional vCRL1 allele most probably would not reveal the function of the protein. Silencing the expression of both vCRL1 alleles, however, promises to be a much more informative approach. Since injection of dsRNA and siRNA into adult Ciona individuals did not cause any detectable downregulation of vCRL1 transcript level (supplementary fig. S7, Supplementary Material online), we developed a technique for tissue-specific RNAi in adult C. intestinalis using stable transgenesis of hairpin constructs. This method was then used to silence vCRL1 expression.
At first, we developed constructs driving reporter expression into all adult tissues or specifically into the blood cell lineage. All constructs were based on the pMiLRneo vector and contained 2 kb of the 5′ flanking sequences of the C. intestinalis EF1α or vWA-like gene (fig. 3A). According to the EST expression data (see Aniseed and Kyoto databases in Tunicate Web Portal http://www.tunicate-portal.org/index.htm), EF1α is expressed in all adult tissues and vWA-like expression is restricted to hemocytes only (detailed analysis in Wakoh et al. 2004). In all constructs, EGFP was used as reporter and 1.5 kb of the 3′ flanking region of EF1α gene insured proper transcription termination and polyadenylation (fig. 3A).
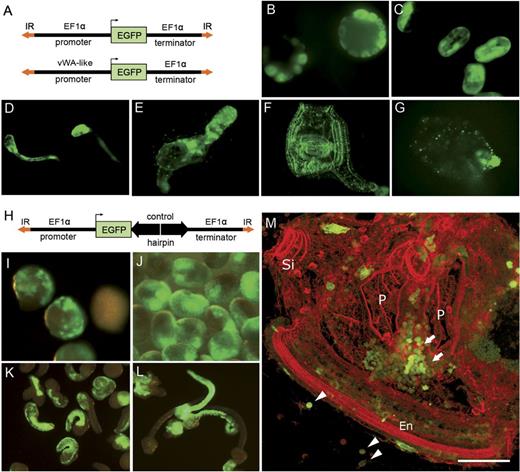
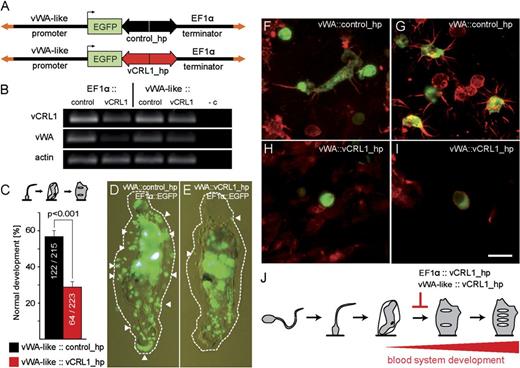
Development of a novel knockdown strategy based on hairpin constructs. (A) Constructs used for transfection utilize EGFP driven by 5′ flanking regions of either EF1α (ubiquitous expression) or vWA-like (hemocyte-specific expression) genes. IR—Minos inverted repeats. EGFP—enhanced green fluorescent protein. (B–F) Fluorescence pictures of Ciona embryos at various developmental stages injected with EF1α∷EGFP promoter construct. (B) Early cleavage, (C) late neurula, (D) late tailbud, (E) rotation, (F) first ascidian stage. EGFP fluorescence is detected in all tissues. (G) First ascidian stage juvenile transfected with vWA-like∷EGFP promoter construct. Only blood cells and tunic cells are GFP positive. (H) Control hairpin construct based on the inverted repeat of Hydra signal transmitter 1 gene. (I–M) Embryos electorporated with the control hairpin construct at early cleavage (I), late gastrula (J), early and late tailbud (K, L), first ascidian stage (M, phalloidin counterstained—red color). Note accumulation of GFP+ blood cells (arrows) between prostigmata near endostyle and GFP+ tunic cells (arrowheads). P—prostigmata, Si—siphon, En—endostyle. Scale bar—50 μm.
Functionality of EF1α∷EFGP and vWA-like∷EGFP reporter constructs has been checked by embryonic microinjection. As shown in figure 3B–F, for the EF1α construct, EGFP fluorescence was detected starting from the early cleavage stages, during larval development, rotation stage, and in all adult tissues. In contrast to that, vWA-like promoter construct was inactive throughout the embryonic development and metamorphosis. EGFP expression was detectable only in hemocytes and tunic cells starting from the first ascidian stage (fig. 3G). Thereby, our results with the vWA-like∷EGFP reporter correspond well to the first appearance of hemocytes at the rotation stage during the transition to the first ascidian stage (Chiba et al. 2004).
As a next step, we generated a control hairpin construct, which contains an inverted repeat of a gene from H. vulgaris AEP cloned between EGFP and EF1α terminator (fig. 3H). Functionality and the influence of the construct on the development have been assessed after electroporation. As shown in figure 3I–L, introduction of a plasmid producing dsRNA did not have any adverse effect on embryonic development in Ciona. Transgenic animals successfully underwent metamorphosis and developed into normal young juveniles within 7 days (fig. 3M). Multiple tissues including blood cells were stably labeled with EGFP (fig. 3M).
Knockdown construct for vCRL1 was produced by cloning parts of the vCRL1 gene in opposite orientations into EF1α∷EGFP plasmid (fig. 4A). 3′UTR was included because it is the only part of the vCRL1 gene, which is highly conserved among Ciona individuals. For knockdown experiments, batches of embryos were electroporated with the control hairpin (EF1α∷control_hp) and vCRL1 hairpin (EF1α∷vCRL1_hp) plasmids (fig. 4B). Only those electroporated animals were chosen for further analysis, which showed more than 75% transgenic cells in the whole embryo and normal tadpole development. Progress of metamorphosis was scored by counting the number of individuals, which succeeded in transition from one stage of the metamorphosis to another (for details, see Materials and Methods).
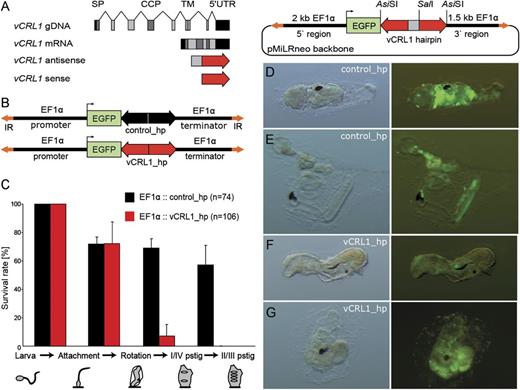
Knockdown of vCRL1 expression effects metamorphosis and blood cell development. (A) Schematic representation of the vCRL1 gene and the corresponding knockdown construct. Complementary parts of the hairpin (antisense and sense) are shown in red, spacer sequence depicted in gray. (B) Schematic representation of control and vCRL1 hairpin constructs driven by EF1α promoter. (C) Percentage of embryos transfected with vCRL1 or control hairpin constructs, which successfully developed through the indicated stage. Data represent the mean of three independent experiments. Knockdown of vCRL1 expression results in developmental arrest at rotation stage. (D, E) Metamorphosis in animals electroporated with control hairpin construct proceeds normally. (F, G) Animals electroporated with vCRL1 hairpin construct start rotation but later round up and do not reach the stage with developed prostigmata.
As shown in figure 4C, about 70% of larvae electroporated with EF1α∷control_hp or EF1α∷vCRL1_hp entered rotation stage. Settlement and tail absorption proceeded normally in the control hairpin (fig. 4D) and vCRL1 hairpin electroporated animals (fig. 4F). However, only 7% of the animals electroporated with EF1α∷vCRL1_hp developed prostigmata compared with 68.9% of animals in the control. In a striking contrast to the control animals (fig. 4E), vCRL1 knockdown animals acquired rounded shape and did not develop further (fig. 4G). None of the embryos transfected with the EF1α∷vCRL1_hp construct completed metamorphosis, whereas development was not impaired by the control hairpin. As shown in figure 4C, metamorphosis in most of the EF1α∷vCRL1_hp animals was arrested at the transition from rotation to the first ascidian stage.
Next, we addressed the question whether the developmental arrest is a systemic effect or is due to the vCRL1 knockdown in hemocytes. For that purpose, we generated transgenic animals where vCRL1 hairpin was expressed under the control of vWA-like promoter, which is hemocyte specific (fig. 5A). vWA-like driven constructs were coelectroporated with EF1α∷EGFP plasmid, which served as a proxy for electroporation efficiency. This approach allowed us to select embryos with at least 75% of transgenic cells already at the tailbud stage—before the first appearance of GFP+ blood cells.
First, we compared functionality of the hairpin constructs driven by EF1α and vWA-like promoters by RT-PCR (figure 5B). Each batch of fertilized embryos was split into four parts and electroporated with the following plasmids: EF1α∷control_hp, EF1α∷vCRL1_hp, vWA-like∷control_hp + EF1α∷EGFP, and vWA-like∷vCRL1_hp + EF1α∷EGFP (see figs. 4B and 5A). mRNA was extracted from the embryos at the rotation stage, and amounts of cDNA taken for RT-PCR were equilibrated based on ß-actin expression. As shown in figure 5B, RT-PCR demonstrates clear depletion of vCRL1 transcript in EF1α∷vCRL1_hp electroporated animals compared with EF1α∷control_hp. In vWA-like∷vCRL1_hp, we also observe considerable depletion compared with vWA-like∷control_hp. RNAi effect is stronger with EF1α∷vCRL1_hp construct than in the vWA-like∷vCRL1_hp construct. The most interesting observation is the drastic decrease in the amount of vWA-like transcript in the vCRL1 knockdown animals (electroporated with EF1α∷vCRL1_hp or vWA-like∷vCRL1_hp constructs). As vWA-like gene is a highly specific blood cell marker in Ciona (see Wakoh et al. 2004), our data, therefore, demonstrate that downregulation of vCRL1 expression strongly influences blood cell population.
Knockdown of vCRL1 expression in the blood cell lineage. (A) Schematic representation of control and vCRL1 hairpin constructs driven by vWA-like promoter. (B) Expression of vCRL1 and vWA-like genes in knockdown animals. RT-PCR with mRNA extracted at rotation stage from animals electroporated with EF1α∷control_hp, EF1α∷vCRL1_hp, vWA-like∷control_hp, and vWA-like∷vCRL1_hp constructs. cDNA amounts were equilibrated using beta-actin. (C) Percentage of embryos transfected with vCRL1 or control hairpin constructs, which successfully developed into first ascidian stage. (D) Animal coelectroporated with vWA∷control_hp + EF1α∷EGFP. Large amount of GFP+ tunic cells are visible at the rotation stage. (E) Animal coelectroporated with vWA∷vCRL1_hp + EF1α∷EGFP. Few GFP+ tunic cells are detectable at the rotation stage. In (D) and (E), GFP+ tunic cells are marked with white arrowheads and tunic border with a dotted line. (F, G) Blood cells and tunic cells are GFP+ in vWA-like∷control_hp transgenic animals. (H, I) Differentiated GFP+ tunic cells were not detected in vWA-like∷vCRL1_hp transgenic animals. Only round-shaped circulating cells (H) or cells with signet ring morphology (I) were present. (J) vCRL1 gene is essential for the establishment of the functional blood system in Ciona. Developmental defects caused by vCRL1 silencing take place at the transition from the rotation to the first ascidian stage when the blood system is normally established. Scale bar (F–I)—10 μm.
After checking RNAi efficiency, we compared the process of metamorphosis in embryos electroporated with vWA-like∷control_hp and vWA-like∷vCRL1_hp. In both cases, fertilized eggs were coelectroporated with EF1α∷EGFP plasmid, which allowed us to select highly transgenic embryos at the tailbud stage. Progress of metamorphosis was scored in three independent experimental groups. As shown in figure 5C, 56.7% of animals (122 of 215) electroporated with vWA-like∷control_hp construct developed into young juveniles (with two prostigmata). At the same time in vWA-like∷vCRL1_hp electroporated animals, only 28.7% (64 of 223) could develop two prostigmata, which is a highly significant difference (χ2 test, P < 0.001). We observed 28% decrease in the efficiency in metamorphosis between vWA-like∷vCRL1_hp construct and corresponding control. Thus, expression of vCRL1 dsRNA only in the blood cell lineage also causes block of metamorphosis at the rotation stage. While following the progress of metamorphosis, we noticed that in the control animals (vWA-like∷control_hp + EF1α∷EGFP), numerous GFP+ tunic cells could be observed during the rotation stage (fig. 5D). In contrast to that, in animals with vCRL1 knockdown (vWA-like∷vCRL1_hp + EF1α∷EGFP), the number of GFP+ cells in tunic was drastically reduced (fig. 5E), which corresponds well to the results of RT-PCR for the expression of vWA-like gene.
Most straightforward explanation for the observed developmental defect is the reduction in the number of differentiated blood cells. This view is also supported by the fact that in vWA-like∷vCRL1_hp transgenic animals (without EF1α∷EGFP tracer) we could not detect GFP+ tunic cells with filopodia, which are differentiation products of circulating hemocytes. As shown in figure 5F and G, in controls, both round-shaped blood cells and large numbers of their GFP+ derivatives—tunic cells with filopodia were present. In contrast to that, in vWA-like∷vCRL1_hp transgenic animals, we detected only GFP+ round-shaped blood cells (fig. 5H) or signet ring cells (fig. 5I).
Taken together, overexpression of vCRL1 dsRNA using EF1α or vWA-like promoters causes similar phenotype—block of metamorphosis at the rotation stage, exactly at the phase when the first hemocytes normally originate (fig. 5J). There is a clear correlation between vCRL1 knockdown and disappearance of a hemocyte-specific marker vWA-like. It seems that in the absence of vCRL1 transcript, blood cell proliferation (or differentiation) is severely disturbed. Thus, our data demonstrate that vCRL1 gene might be essential for the establishment and maintenance of a functional blood system in C. intestinalis.
Discussion
vCRL1 is an interesting gene because of three reasons. First, it is extraordinarily polymorphic (Kürn et al. 2007). Every animal carries two vCRL1 alleles, which are unique—in their sequence, they differ from any allele of any other randomly sampled individual. So far, we have never detected identical vCRL1 alleles unless two animals were closely related, being, for example, siblings from experimental crosses. Polymorphism of the gene is extremely high—the average similarity between randomly sampled alleles varies between 70% and 93% on the protein level (Kürn et al. 2007). Such a variability is the hallmark of genes involved in allorecognition as it allows to distinguish one conspecific individual from another. Second, vCRL1 is expressed in tissues (follicle cells and hemocytes), which according to several publications are responsible for self/nonself discrimination in Ciona. Follicle cells have been proposed to play a role in preventing selfing (Rosati and de Santis 1978; De Santis et al. 1979; De Santis and Pinto 1991) and hemocytes take part in the rejection of tunic allografts (Reddy et al. 1975). Third, vCRL1 is 1 of the 132 genes in Ciona, which according to their domain organization belong to putative regulatory components of the complement system (Azumi et al. 2003). Number of genes with SCR/CCP domains has more than doubled in Ciona lineage compared with 53 genes in human (Azumi et al. 2003) and their function remained unknown.
Initially, we investigated whether vCRL1 is a self-sterility receptor. In Ciona, there are two subspecies—A and B, which inhabit Pacific and North Atlantic, respectively (Suzuki et al. 2005). In subspecies A, two loci responsible for self-sterility have been previously identified by Harada et al. (2008). In our experiments, we used subspecies B of C. intestinalis from North Atlantic. Degree of the genetic variation between subspecies A and B is such that animals belonging to different “types” do not interbreed and according to the classical definition might even be regarded as different species (Caputi et al. 2007). Thus, it was reasonable to assume that subspecies A and subspecies B are already on the way to develop different molecular mechanisms to prevent selfing. Our segregation analysis shows that vCRL1 gene is not a self-sterility receptor. At the same time, it clearly demonstrates that in subspecies B, Themis loci are also utilized in order to prevent self-fertilization (fig. 2). Thereby, the same genes block self-fertilization in two reproductively isolated subspecies of C. intestinalis. It means that receptors and ligands that ensure species-specific recognition between gametes and Themis genes, which prevent self-fertilization, function autonomously and seem to evolve independently.
Our experiments demonstrate that vCRL1 gene is not involved in self-sterility but is essential for blood system formation in Ciona. If we knock down vCRL1 expression throughout the body or specifically in the blood cell lineage—metamorphosis is blocked, and the amount of hemocytes is drastically reduced (figs. 4 and 5). In the present work, we did not analyze the downstream molecular effects of vCRL1 knockdown. That surely needs further investigation. However, we would like to propose the hypothetical model, which explains the observed phenotype. Taking into consideration the individual-specific nature of vCRL1 polymorphism, we think that this protein may function as a hemocyte-specific self marker. Recognition system based on vCRL1 locus might be responsible for self/nonself discrimination reactions among blood cells within the organism (fig. 6A) and might interact with the effector arm of the complement system, which is highly developed in Ciona (Azumi et al. 2003; Nonaka and Kimura 2006). In tunicates, lysis between allogeneic blood cells termed “contact reaction” is well documented for H. roretzi (Fuke and Numakunai 1982; Fuke 2001), and allogeneic cytotoxicity has been reported in Styela clava (Kelly et al. 1992). In Ciona, this phenomenon has not been reported. In contrast to Halocynthia, hemocytes of Ciona possess an extremely high capacity for immediate agglutination. With high concentrations of chelating agents (e.g., ethylenediaminetetraacetic acid), formation of cellular aggregates can be prevented, but this treatment most probably strongly influences all the interactions between blood cells.
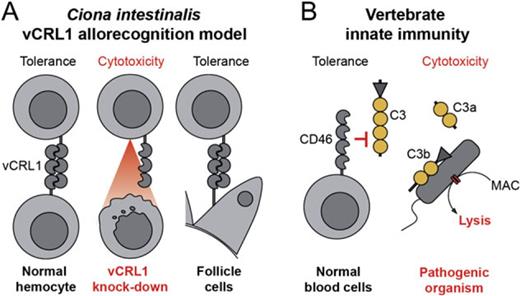
Molecular mechanisms of self/nonself recognition in Ciona intestinalis. (A) Hypothetical model of somatic self/nonself recognition in C. intestinalis mediated by vCRL1 proteins. All hemocytes express individual-specific vCRL1 gene, which marks them as “self.” Binding of soluble complement components or direct lysis by other hemocytes is thereby inhibited. Foreign cells, which do not carry such a self-marker, are destroyed by default. As a result of the vCRL1 knockdown, hemocytes could become autoreactive or may be lyzed by the complement system since the appropriate self-marker is missing. That leads to developmental arrest at the transition from rotation to first ascidian stage. Presence of vCRL1 at the surface of follicle cells may be necessary for the protection against one's own immune system during accumulation of mature oocytes in oviduct. (B) “Missing-self” strategy of innate immunity exemplified by the mammalian complement system. Soluble complement factors cannot attach to the plasma membrane of cells, which carry protective complement receptors (CD46, CD55, etc.). Thereby, these proteins inhibit binding and processing of complement components and the formation of pores. MAC—membrane attacking complex.
How can vCRL1 receptor allow discrimination between self and nonself? We propose that vCRL1 might be an inhibitory receptor, which protects cells of the “own” tissue by labeling them as self and, thereby, preventing lysis by the complement system or by other hemocytes (fig. 6A). If cells are not carrying vCRL1 on their surface, either because of their allogeneic origin (nonself) or due to knockdown by RNAi, they are not recognized as self, and tolerance is not established. Instead, these cells are attacked by immune system and destroyed. Consequently, animals deprived of vCRL1 expression in hemocytes are not able to establish a functional blood system during metamorphosis. This then results in an arrest of development at the transition from rotation stage to first ascidian stage (figs. 4 and 5) exactly at the moment when the first circulating hemocytes normally emerge (Chiba et al. 2004). Our hypothesis also explains why vCRL1 is needed on the surface of follicle cells, which cover oocytes. In Ciona, mature oocytes accumulate in oviduct, which connects the ovary with the surrounding seawater. If vCRL1 functions as an inhibitory receptor, it might be used to protect oocytes from the innate immune system of the animal during their storage prior to spawning.
vCRL1 gene encodes a protein with a structural similarity to human complement receptors CD46 and CD55, which regulate the activation of the complement pathway. Serving as self-marker for innate immune system, they protect cells from lysis by own complement system. All cells with CD46 and CD55 on the surface are referred to as self, and every cell without these receptors is opsonized and destroyed (fig. 6B). We suggest that the mode of vCRL1 function in Ciona might be very similar to that of CD46 and CD55 in human. However, in contrast to mammalian complement control proteins, vCRL1 is highly polymorphic and individual specific. Why should self-marker vary in the population of solitary animals? In mammals, for example, NK receptors, which function according to the “missing-self” mechanism, are not polymorphic (Kärre 2002). We propose that using individual-specific and highly variable receptors as a self-marker makes it possible to counteract any adaptive coevolution of pathogenic organisms, not letting them to evade immune response of the host. Such a system has a crucial advantage over all nonpolymorphic receptors or pathogen-associated molecular pattern sensors. It would facilitate identification of any possible intruder as nonself no matter to which taxonomic group the pathogen belongs.
Taken together, functional analysis of vCRL1 indicates that receptors with SCR/CCP domains might have an evolutionary ancient function not only in regulation of innate immunity but also in self/nonself recognition. The fact that the histocompatibility locus (Fu/HC) of Botryllus encodes highly polymorphic protein with SCR/CCP unit also points in this direction (Nyholm et al. 2006). It would be of great interest to investigate whether other groups of invertebrate animals possess variable receptors with SCR/CCP domains.
We are grateful to the staff of the Biological Station on Helgoland for supplying us with C. intestinalis and to Antje Thomas for help with histological techniques. Furthermore, we are thankful to Sebastian Fraune and Sören Franzenburg for providing the control hairpin construct and to Georg Hemmrich for help with assembly of bacterial artificial chromosome (BAC) clones. We thank Santiago Insua for IT and bioinformatics support. This work is supported by grants from the Deutsche Forschungsgemeinschaft (DFG, http://www.dfg.de) to K.K. and T.C.G.B. and grants from the DFG Cluster of Excellence programs “The Future Ocean” and “Inflammation at Interfaces.”
References
Author notes
Associate editor: Billie Swalla